
Contact information

Paul Holloway
Principal Investigator
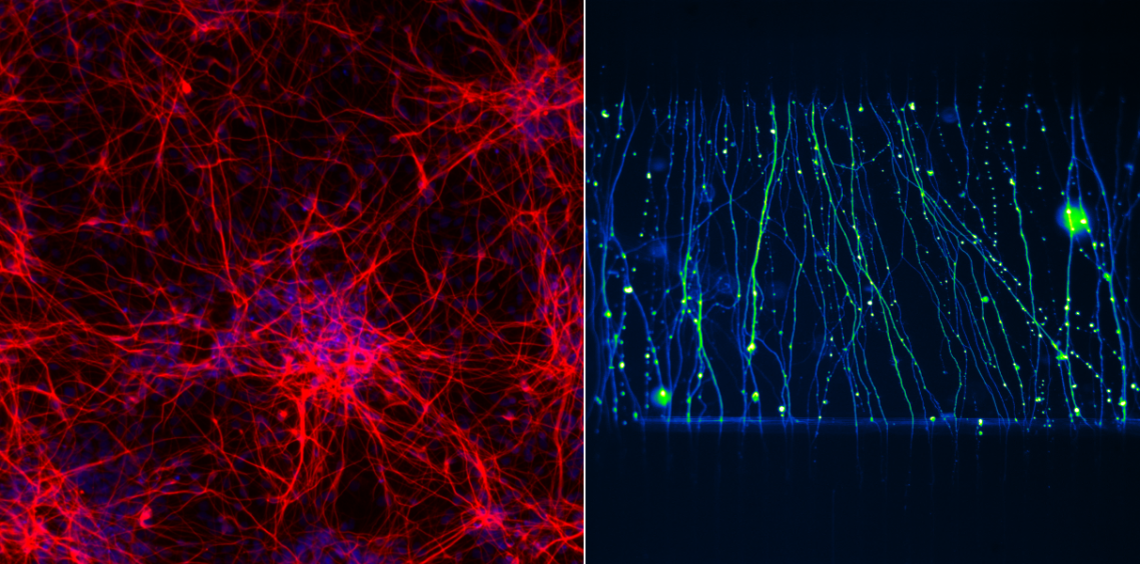
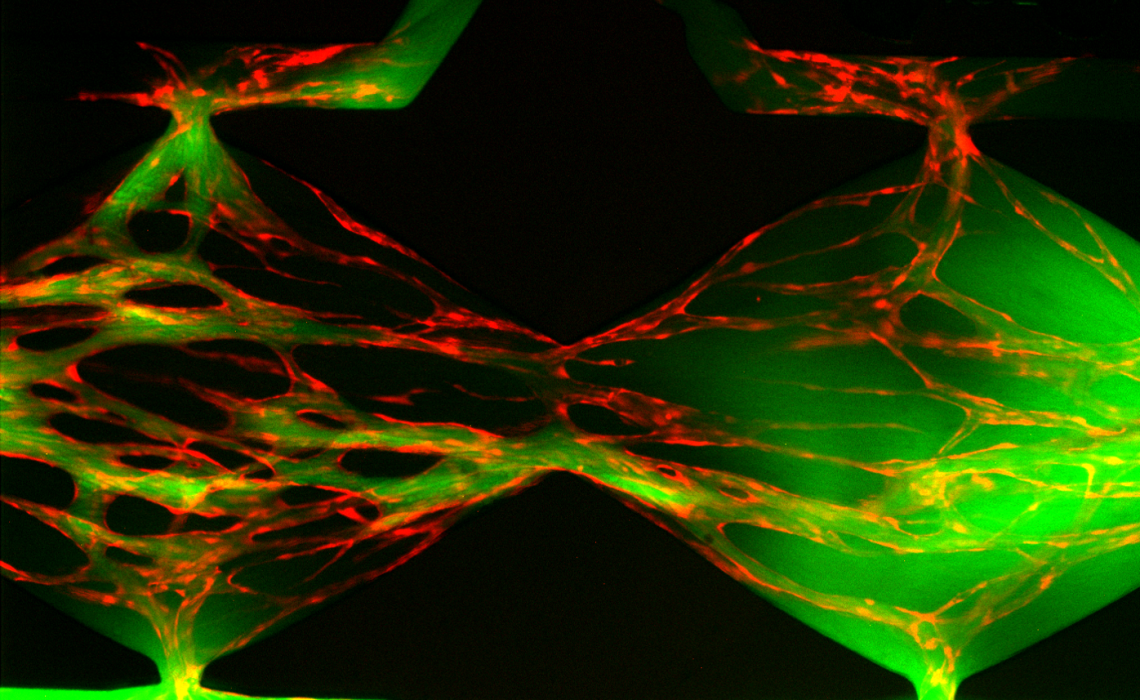
My research utilises microfluidic technologies to precisely define the cellular micro-environment and enable new insights into neurovascular biology.
In the brain, endothelial cells, perivascular cells, glia and neurons are intimately coupled, enabling tight regulation of the blood brain barrier, responses to metabolic demands, and inflammatory status. This has lead to the consideration of these cellular elements as part of a functional unit termed the 'neurovascular unit'. Disruption of this system is evident in many human neurological disorders. However, investigating this system in humans and modelling the complex interactions in vitro is particularly challenging.
Recently, micro-fabrication techniques adopted from the semiconductor industry have allowed the production of miniaturised biological devices for complex, spatially-defined cell culture which can be used to mimic human organs. As part of my Royal Commission Research Fellowship, at the University of Oxford’s Laboratory of Cerebral Ischemia, I am utilising ‘Organ on chip’ technologies to develop a three-dimensional, dynamic, heterotypic cellular model of the neurovascular unit. The pathophysiological relevance of this model will be interrogated in the context of stroke, testing the fidelity of cellular coupling and interaction by metabolically perturbing the system.
Biography
Dr Holloway has a background incorporating both neurovascular biology and microfluidic engineering. His PhD at Imperial College London and Post-doctoral position at Louisiana State University focused on novel therapeutic approaches to modulate immune cell trafficking in the cerebral microcirculation following stroke. Initial investigations using minimalistic flow assays, to investigate human immune-brain cell interactions in vitro, led Dr Holloway to pursue training in microfluidic engineering approaches at Princeton University followed by a post-doctoral position at the Centre for Hybrid Biodevices, University of Southampton. In this position Dr Holloway helped develop compartmentalised microfluidic cell culture platforms for the preparation of defined neuronal circuits to shed light on axonal transport and disease propagation. His current work utilises both advanced cell culture and microfabrication techniques to develop an organ on chip model of the neurovascular unit.
